|
Images |
|
|
|
|
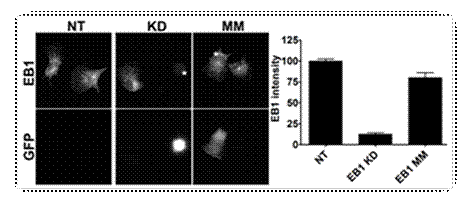
Knock
down of EB1 using short hair-pin RNA interference. B16F10 cells were transfected
with EB1 knock down (KD) or MM control plasmids and stained with anti-EB1
monoclonal antibodies. Non-transfected
(NT) cells serve as an additional control. GFP co-expression identifies cells
transfected with EB1 KD or MM control plasmids (lower panel). Whole cell EB1
fluorescence intensities are plotted relative to NT cells. |
|
|
|
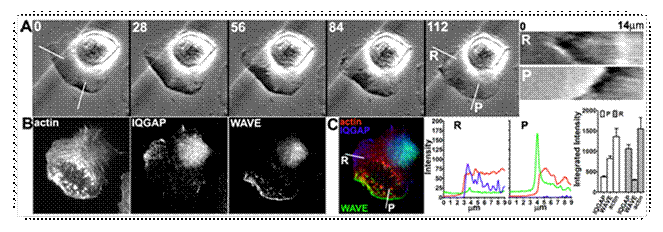
IQGAP and WAVE localization at retracting and protruding
edges. (A)
B16F10 cells were plated on laminin for 30 minutes then phase images were
acquired at the indicated times (0-122 seconds). Kymographs represent
retracting (R) and protruding (P) cell edges. (B) Cells were
immediately fixed following live cell imaging and stained for actin, IQGAP1 and
WAVE2. (C) Fluorescence intensity of actin (red), IQGAP1 (blue) and WAVE2
(green) at retracting and protruding edges are plotted. |
|
|
|
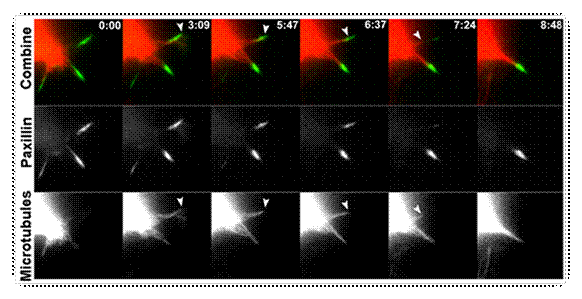
Microtubule targeting
precedes dissociation of adhesions sites in B16F1 cells. Cells
were co-transfected with GFP-paxillin and monomeric RFP-beta-tubulin subunit
(mRFP-tubulin), and plated onto laminin.
Time is given in minutes and seconds. Arrows indicate a microtubule
end targeting an adhesion site marked by GFP-paxillin. |
|
|
|
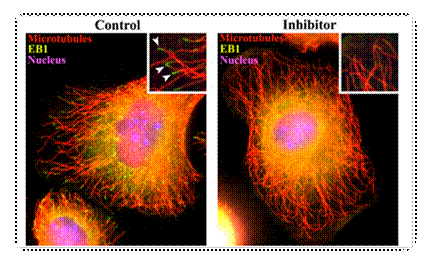
Polymerization is
required for EB1 association to microtubule plus-ends. Left: In
control samples EB1 binds to the plus-ends of microtubules (inset, arrows). Right: In the presence of a known
inhibitor (nocodazole for 30 sec) of microtubule polymerization EB1 binding
to microtubule plus-ends is inhibited. |
|
|
|
A moving mouse
melanoma cell. The cell was fixed and stained for
actin. Note numerous filopodia along
the cell front. |
|
|
|
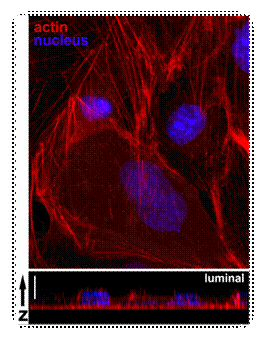
Actin and nuclei in
endothelial cells with z-stack. A
monolayer of endothelial cells was stained for actin and DNA. The z-series of
images was deconvoluted before assembly into stack. |
|
|
|
SIUe
School of Pharmacy Pharmaceutical Sciences Doctor of Pharmacy Degree Biology Research Schober lab Flickr |
|
|
|
The views and opinions expressed in this page are
strictly those of the page author. The contents of this page have not been
reviewed or approved by SIUE. |
|